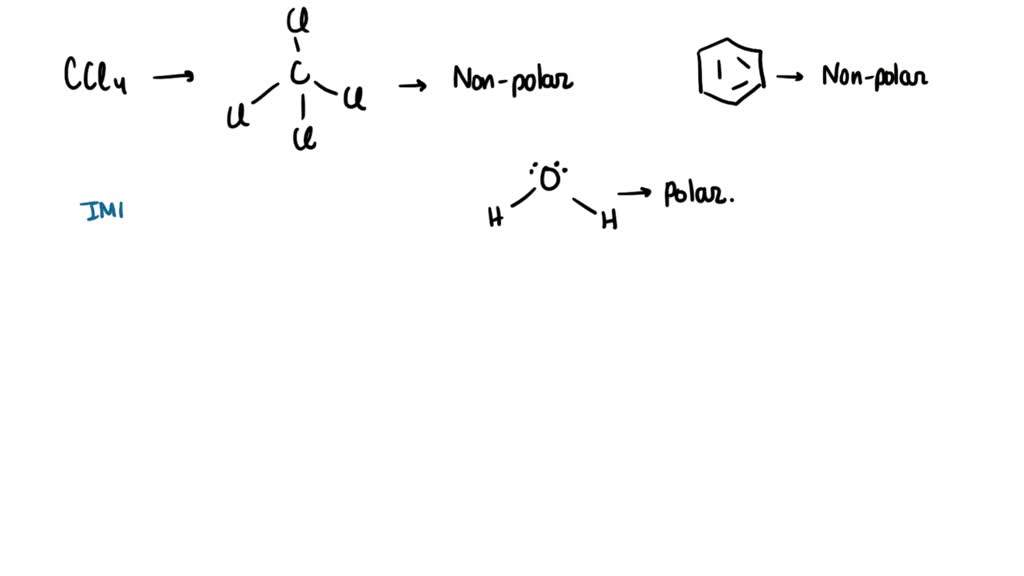
text{ }{{\\text{C}}_{\\text{6}}}{{\\text{H}}_{\\text{6}}}\\text{ }$ is a very good industrial solvent for:A) $\\text{ NaCl }$B) $\\text{ MgC}{{\\text{l}}_{\\text{2}}}\\text{ }$C) $\\text{ CaC}{{\\text{O}}_{\\text{3}}}\\text{ }$D) Fats

SOLVED:Water is a polar solvent and benzene is a nonpolar solvent. In which solvent is each of the following solutes more likely to be soluble? a. hexane b. gasoline c. KCl d.